NOMS Audiology Registry
NOMS is a voluntary data collection registry that illustrates the value of audiological services and enables clinicians to improve the quality of those services.
The key to the NOMS Audiology Registry is the use of a combination of patient-reported outcomes (PROs) and clinically-administered tests to capture clinical improvements before and after audiological intervention.
To participate in the NOMS Audiology registry, audiologists must be licensed to practice in the state in which services are provided. The registry is provided at no cost to ASHA members. Nonmembers can participate by purchasing a NOMS Registry Subscription.
Data Collection
NOMS data collection is quick, is easy, and involves the reporting of information already included in the patient's record. This includes information on the following:
- Demographics
- Diagnosis and CPT codes
- Functional status using condition-specific patient-reported outcomes
- Assessment and treatment information
Audiologists submit patient records to NOMS at the time of initial evaluation when treatment recommendations are made and at regular milestone appointments, including annual evaluations and following the initial acclimatization period of amplification, to track progress over time.
You will be asked to collect data on all patients ages 16 years and older who have received a comprehensive hearing test (hearing condition), tinnitus evaluation (tinnitus condition), and/or vestibular assessment/canalith repositioning (vestibular condition) and treatment recommendations have been made.
For NOMS purposes, you must evaluate at least one condition for each submission—hearing, tinnitus, and vestibular.
- If you evaluate the hearing condition, you should administer the Revised Hearing Handicap Inventory (RHHI) and/or the Abbreviated Profile of Hearing Aid Benefit (APHAB) PRO. Other data elements collected for the hearing condition include ABR stimulus, Speech-in-Noise Testing (CNC-Words or SNR-50), Aural Rehabilitation, and treatment recommendations. This aligns with the core outcome set identified by the NASEM report.
- If you evaluate the tinnitus condition, you should administer the Tinnitus Handicap Inventory (THI) PRO. You will also capture treatment recommendations.
- If you evaluate the vestibular condition, you should only include patients who have received a vestibular assessment and/or canalith repositioning. Instead, of a PRO, enter which maneuvers were performed and how many times each maneuver was administered.
Once you've registered to participate in NOMS, you can submit data in one of two ways:
- By manual entry via our web-based tool
- By electronic transmission directly from your organizations internal documentation system.
You will not be asked to collect any protected health information (e.g., name, date of birth) or withhold treatment in any way.
Data Reports
As a NOMS participant, you will have access to a NOMS dashboard that provides an in-depth analysis of your organization’s outcomes benchmarked against the national data. The interactive dashboard allows you to generate customizable data reports that can help you and your organization improve the quality of your care and demonstrate the effectiveness of audiology services.
The reports include a number of features:
- Interactive dashboards with visualizations in graphical or tabular views
- Greater flexibility for analyzing your data using various filters
- Organization-level benchmarks (if applicable) combined with national benchmarks
- Option to save report templates
- Ability to schedule your reports to run at a future date
- Choice to download the report in PDF or CSV file format
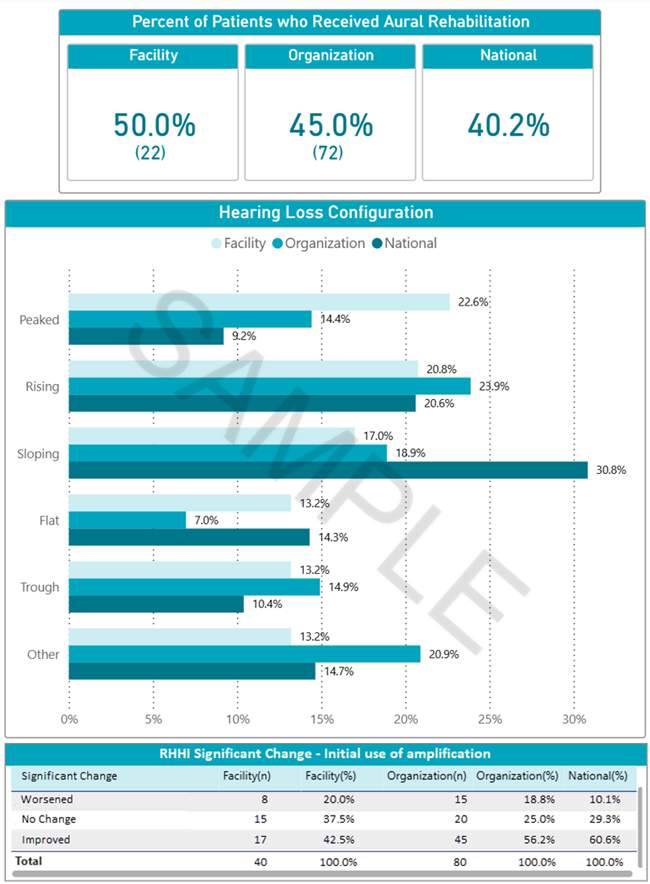
NOMS Audiology Registry report sample.
Contact NOMS@asha.org with any questions.










